We use Faraday’s constant to calculate the amount of electric charge that flows through a circuit per unit of time. The Faraday constant is equal to the electric charge per mole of elementary charges (like electrons). One mole is defined as the number of atoms or molecules in exactly 12 grams of pure carbon-12. This number is called Avogadro’s number (NA) and is approximately 6.022 x 1023 atoms or molecules.
What Is the Faraday Constant in Mathematical Terms?
F = NA*e
Where:
F= Faraday’s constantNA= Avogadro’s constant (approximately6.022 x 1023electrons per mole)e= the charge of a single electron (approximately1.602 x 10-19Coulombs)
So, we define the Faraday constant as the charge of one mole of electrons, which is equal to Avogadro’s number (NA) of electrons.
How Does the Faraday Constant Work?
To understand how Faraday’s constant works, let’s look at how an electrolytic cell works.
A typical electrolytic cell consists of two electrodes submerged in an electrolyte solution. When we apply an electric current to the electrodes, one electrode acts as an anode and the other electrode acts as a cathode. At the anode, oxidation takes place and the anode loses electrons. At the cathode, reduction takes place and the cathode gains electrons. The electrons lost at the anode flow through the external circuit to the cathode where they are gained. This process is depicted in the figure below:
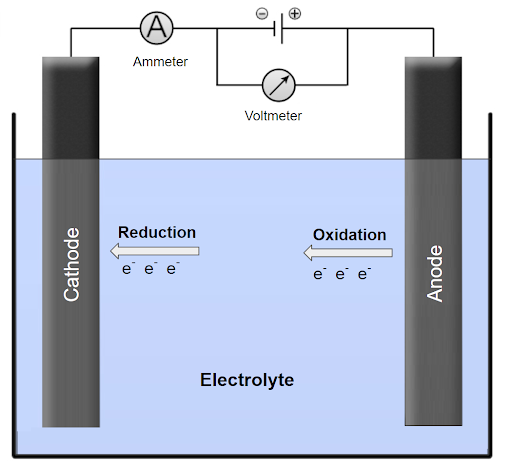
The amount of electric charge passed through the cell is related to the amount of substance that’s being oxidized or reduced at the electrodes.
Faraday’s constant relates the amount of electric charge passed through the cell (Q) to the amount of substance involved in the reaction (n) by the following equation:
Q = nF
Here, Q is the electric charge passed through the cell, n is the amount of substance involved in the reaction and F is Faraday’s constant.
The value of Faraday’s constant is fixed at 96,485.3399 Coulombs per mole of electrons, which means that one mole of electrons is equivalent to 96,485.3399 Coulombs of electric charge.
We can use this equation to calculate the amount of substance involved in the reaction by measuring the electric charge passed through the cell and multiplying it by the reciprocal of Faraday’s constant.
Why Is the Faraday Constant Important?
Faraday’s constant is a fundamental constant in science that we use to understand and quantify the amount of electric charge involved in many chemical and physical phenomena.
Applications of the Faraday Constant
We use the Faraday constant in a variety of fields from electrochemistry to thermodynamics, battery technology, analytical chemistry and more.
Electrochemistry
We use Faraday’s constant in electrochemistry to calculate the amount of electric charge that is involved in a chemical reaction, as well as to calculate the electrochemical equivalent of a substance. We use this constant to determine the relationship between the amount of electric charge passed through an electrolytic cell and the amount of material that’s electrodeposited or dissolved at the electrodes.
Electrochemical Cells
We use Faraday’s constant in the theoretical study of electrochemical cells to calculate the potential difference between the electrodes and the amount of material that’s involved in the redox reactions.
Thermodynamics
In thermodynamics, we use Faraday’s constant to calculate the Gibbs free energy of electrochemical reactions, which is important in helping us understand the thermodynamics of electrochemical systems.
Battery Technology
Faraday’s constant is important in determining a battery’s capacity. We use the constant in the calculation of the amount of charge required to produce one mole of a substance, which is crucial in determining the amount of energy stored in a battery.
Analytical Chemistry
In analytical chemistry, we use Faraday’s constant in many different techniques like voltammetry, to calculate the amount of substance involved in an electrochemical reaction. We can then use this calculation to detect and quantify the concentration of different species in solutions.
Frequently Asked Questions
What is the Faraday constant?
The Faraday constant is a physical constant equivalent to the amount of electric charge per mole of electrons or another elementary charge. The exact value of Faraday’s constant is 96,485.3399 Coulombs per mole of electrons.
How is the Faraday constant calculated?
Faraday’s constant is calculated with the equation F = NA*e, where NA is Avogadro’s constant and e is the electric charge of one electron.
Why is the Faraday constant important?
The Faraday constant is used to calculate the amount of electric charge in various situations spanning analytical chemistry, thermal dynamics and electrochemistry, among other sectors.





